Acer forrestii
Sponsor
Kindly sponsored by
a member of the International Dendrology Society
Credits
Article from Bean's Trees and Shrubs Hardy in the British Isles
Recommended citation
'Acer forrestii' from the website Trees and Shrubs Online (treesandshrubsonline.
Genus
- Acer
- Sect. Macrantha
Synonyms
- Acer pectinatum subsp. forrestii (Diels) E. Murray
Infraspecifics
Other taxa in genus
- Acer acuminatum
- Acer amplum
- Acer argutum
- Acer barbinerve
- Acer buergerianum
- Acer caesium
- Acer calcaratum
- Acer campbellii
- Acer campestre
- Acer 'Candy Stripe'
- Acer capillipes
- Acer cappadocicum
- Acer carpinifolium
- Acer 'Cascade'
- Acer caudatum
- Acer ceriferum
- Acer chapaense
- Acer chienii
- Acer circinatum
- Acer cissifolium
- Acer × conspicuum
- Acer cordatum
- Acer coriaceifolium
- Acer × coriaceum
- Acer crataegifolium
- Acer davidii
- Acer diabolicum
- Acer distylum
- Acer divergens
- Acer duplicatoserratum
- Acer elegantulum
- Acer erianthum
- Acer 'Esk Flamingo'
- Acer fargesii
- Acer fenzelianum
- Acer flabellatum
- Acer franchetii
- Acer × freemanii
- Acer fulvescens
- Acer 'Gimborn'
- Acer ginnala
- Acer glabrum
- Acer 'Gold Coin'
- Acer granatense
- Acer grandidentatum
- Acer griseum
- Acer heldreichii
- Acer henryi
- Acer × hillieri
- Acer hookeri
- Acer hyrcanum
- Acer japonicum
- Acer kawakamii
- Acer komarovii
- Acer laevigatum
- Acer laurinum
- Acer laxiflorum
- Acer lobelii
- Acer longipes
- Acer macrophyllum
- Acer mandshuricum
- Acer maximowiczianum
- Acer maximowiczii
- Acer metcalfii
- Acer miaotaiense
- Acer micranthum
- Acer 'Mindavi'
- Acer 'Minorient'
- Acer miyabei
- Acer miyabei × campestre
- Acer monspessulanum
- Acer morifolium
- Acer 'Mozart'
- Acer oblongum
- Acer obtusifolium
- Acer okamotoanum
- Acer oliverianum
- Acer opalus
- Acer orientale
- Acer palmatum
- Acer papilio
- Acer pauciflorum
- Acer pectinatum
- Acer pensylvanicum
- Acer pentaphyllum
- Acer pentapotamicum
- Acer pictum
- Acer pilosum
- Acer pinnatinervium
- Acer platanoides
- Acer platanoides × amplum
- Acer platanoides × truncatum
- Acer × pseudoheldreichii
- Acer pseudoplatanus
- Acer pseudosieboldianum
- Acer pubinerve
- Acer pycnanthum
- Acer rubescens
- Acer rubrum
- Acer rufinerve
- Acer saccharinum
- Acer saccharum
- Acer sempervirens
- Acer 'Serpentine'
- Acer serrulatum
- Acer shenkanense
- Acer sieboldianum
- Acer sikkimense
- Acer 'Silver Cardinal'
- Acer 'Silver Ghost'
- Acer sinense
- Acer sinopurpurascens
- Acer spicatum
- Acer stachyophyllum
- Acer taronense
- Acer tataricum
- Acer tegmentosum
- Acer tenellum
- Acer tetramerum
- Acer tibetense
- Acer tonkinense
- Acer triflorum
- Acer truncatum
- Acer tschonoskii
- Acer turkestanicum
- Acer tutcheri
- Acer ukurunduense
- Acer velutinum
- Acer wardii
- Acer 'White Tigress'
- Acer wilsonii
- Acer × zoeschense
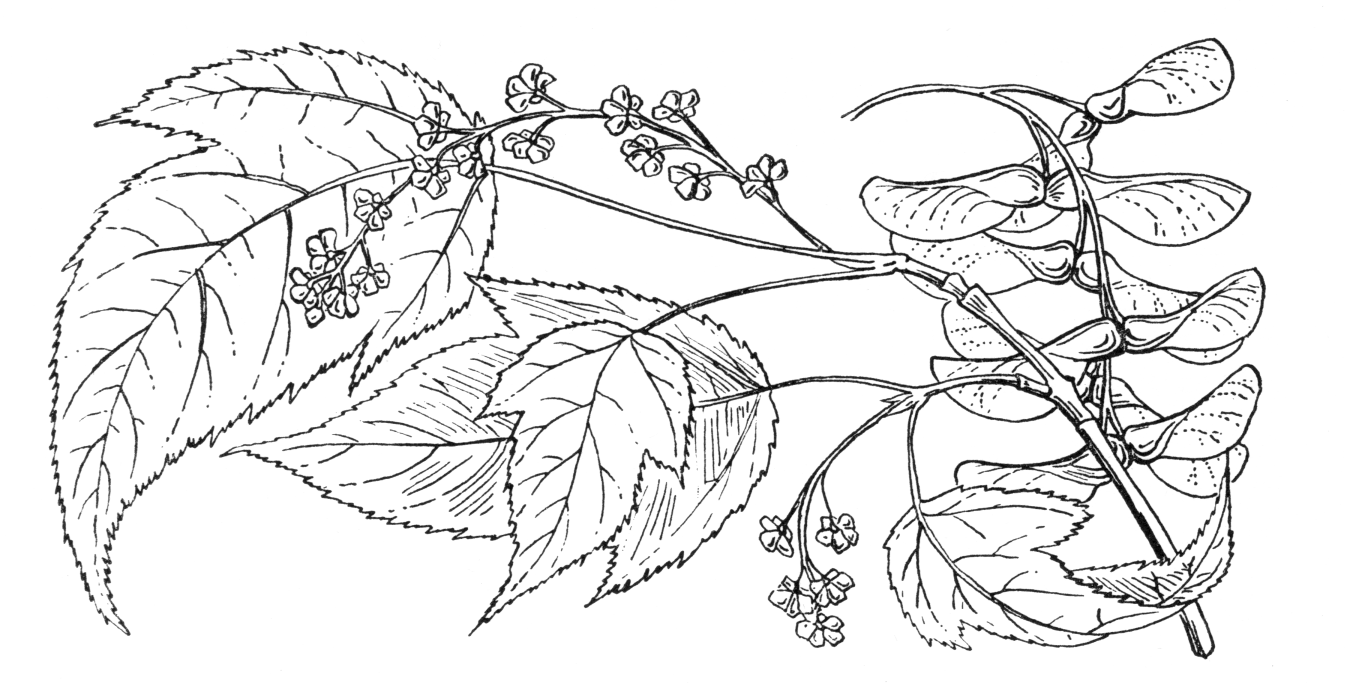
A deciduous tree to 17 m in the wild. Bark dark green with pale stripes. Branchlets glabrous, greenish to purplish-red, faintly striped white. Buds stipitate, small, ellipsoid, glabrous, with 2 pairs of valvate scales. Leaves ovate, chartaceous, base cordate to subcordate, unlobed to 3-lobed, 7–12 × 5–9 cm, central lobe ovate, triangular-ovate, apex caudate-acuminate, lateral lobes triangular-ovate, apex acuminate; margins double-serrulate, teeth adpressed, upper surface dark green, lower surface paler, glaucescent, with pubescent vein axils; petiole 2.5–5 cm long, red or green, grooved, glabrous or pubescent; autumn colours yellow to red. Inflorescence terminal, racemose, pendulous, 5–20 flowered. Flowers yellowish-green, 5-merous, pedicels slender, 0.6–0.8 cm long, sepals oblong, ~0.3 cm long, petals obovate, 0.3–0.4 cm long, stamens 8, inserted outside the nectar disc. Samaras 2.3–2.5 cm long, wings narrowly spreading. Flowering in May, fruiting in October. (Fang 1939; Xu et al. 2008).
Distribution China south western Sichuan, north western Yunnan
Habitat Temperate mixed forests and valleys between 3000 and 3800 m.
USDA Hardiness Zone 6-7
RHS Hardiness Rating H6
Conservation status Least concern (LC)
Taxonomic note The taxonomic position of Acer forrestii has long been a source of debate among acerologists. Treated as subspecies of A. pectinatum by van Gelderen et al. (1994), various authors have also debated its affinity to the taxon treated here as A. laxiflorum, which in turn sits within the broader complex of species allied to A. pectinatum. A comprehensive molecular investigation is needed to resolve many of the issues within this complex, but for now the treatment adopted by Xu et al. (2008) in the Flora of China is followed here. See also the taxonomic notes under A. pectinatum and A. laxiflorum.
The taxonomic position of Acer forrestii has long been a source of debate among acerologists. Introduced by Forrest from Yunnan in 1906, and described from his material, it was reduced to the synonymy of A. laxiflorum by Rehder in 1917 (Sargent 1917), then resurrected as a species by the same author in 1933 (Rehder 1933), this treatment being upheld by Fang (1939) in his monograph of Chinese maples.
Confusingly, in his account of the species and in relation to A. laxiflorum, Bean (1976a) states that ‘there seems to be no other reliable character by which they may be distinguished..’, while in his account of Acer laxiflorum he writes that ‘there should be no possibility of confusing A. laxiflorum with A. forrestii, which has strongly three-veined and three-lobed leaves, glabrous beneath’ (Bean 1976a). While this identification feature is useful, it is too much of a sweeping statement by far, as all manner of intermediates between the two can be found, sometimes on the same plant. Rehder (1933) considered characters of pubescence on lower leaf surfaces in A. laxiflorum and glaucescent undersides in A. forrestii as the most useful way to split the two, though these characters do not always correlate with one another.
van Gelderen et al. (1994) treated Acer forrestii and A. laxiflorum as distinct entities, though as subspecies of A. pectinatum, a treatment first proposed by Murray (1977) and since rejected by Xu et al. (2008) who consider both to be species. However, given the prevelance of intermediate forms between A. forrestii and A. laxiflorum in both leaf characters and flower number, and while it would be rash to jump to any conclusions without the benefit of a comprehensive molecular study, it is reasonable to suggest that this so-called species might sit better within A. laxiflorum, as Rehder had indeed earlier considered (Sargent 1917).
Less debatable is that plants of the Acer forrestii affinity are of great ornamental value. While its autumn colour is unspectacular, its appearance in high summer is anything but. Vibrant red fruits contrast with bright green leaves most effectively. This said, not all examples exhibit this feature, with some origins producing fruit that remains largely green. Among the best red-fruited specimens are those of ACE 2066, ACE 2076 and Clark 4200, the former collected in the Habashan area of Yunnan. All grow at Hergest Croft, Herefordshire, while a plant from ACE 2066 is one of the best for the time of year for any tree at Westonbirt. Also growing in Silk Wood at Westonbirt is a plant of Ogisu 95093 which is also notable for this character. A second plant of ACE 2066 has been added to the Westonbirt collection, acquired from Windsor Great Park, which had propagated plants of it on instruction from the late Mark Flanagan, shortly before his untimely death in 2015. While leaf lobing is not a consistent feature of these plants, particularly on fruiting branches, the greater number of fruits and their glaucous leaf undersides distinguish them from A. laxiflorum (sensu Xu et al. 2008). This applies also to SICH 1461, which forms a broad spreading tree with arching branches on a single stem at Westonbirt. Though grown in some collections as A. maximowiczii, SICH 1059 belongs here. F 30361 was represented at Caerhays, though had died before 2006 (O. Johnson, pers. comm. 2020).
'Alice'
RHS Hardiness Rating: H6
A form with shallowly lobed leaves that are deep pink when young, developing cream and pink variegation. Raised and introduced by Plantentuin Esveld in 1981, having been selected from seedlings grown from seed collected at Westonbirt, UK. It was named after the late Alice van Tright. (van Gelderen et al. 1994; van Gelderen & van Gelderen 1999).
'Sirene'
RHS Hardiness Rating: H6
Raised from a seed and collected from a tree at Plantentuin Esveld, ‘Sirene’ was introduced by the same nursery in 1988 and forms an open branched shrub or small tree. It is darker in all its parts than typical Acer forrestii, while its autumn colour can be reddish orange, though this should not be relied upon. (van Gelderen et al. 1994; van Gelderen & van Gelderen 1999).
'Sparkling'
RHS Hardiness Rating: H6
A more or less upright tree with distinctly lobed leaves and fiery-red petioles, named and introduced by Plantentuin Esveld in 1989 (van Gelderen & van Gelderen 1999).