Davidia involucrata
Sponsor
Kindly sponsored by
Caroline Boisset
Credits
Julian Sutton (2019)
Recommended citation
Sutton, J. (2019), 'Davidia involucrata' from the website Trees and Shrubs Online (treesandshrubsonline.
Genus
Common Names
- Dove Tree
- Handkerchief Tree
- Pocket Handkerchief Tree
- Ghost Tree
- Davidia
- Laundry Tree
Synonyms
- Davidia laeta Dode
- Davidia vilmoriniana Dode
- Davidia involucrata var. vilmoriniana (Dode) Wangerin
Infraspecifics
Other taxa in genus
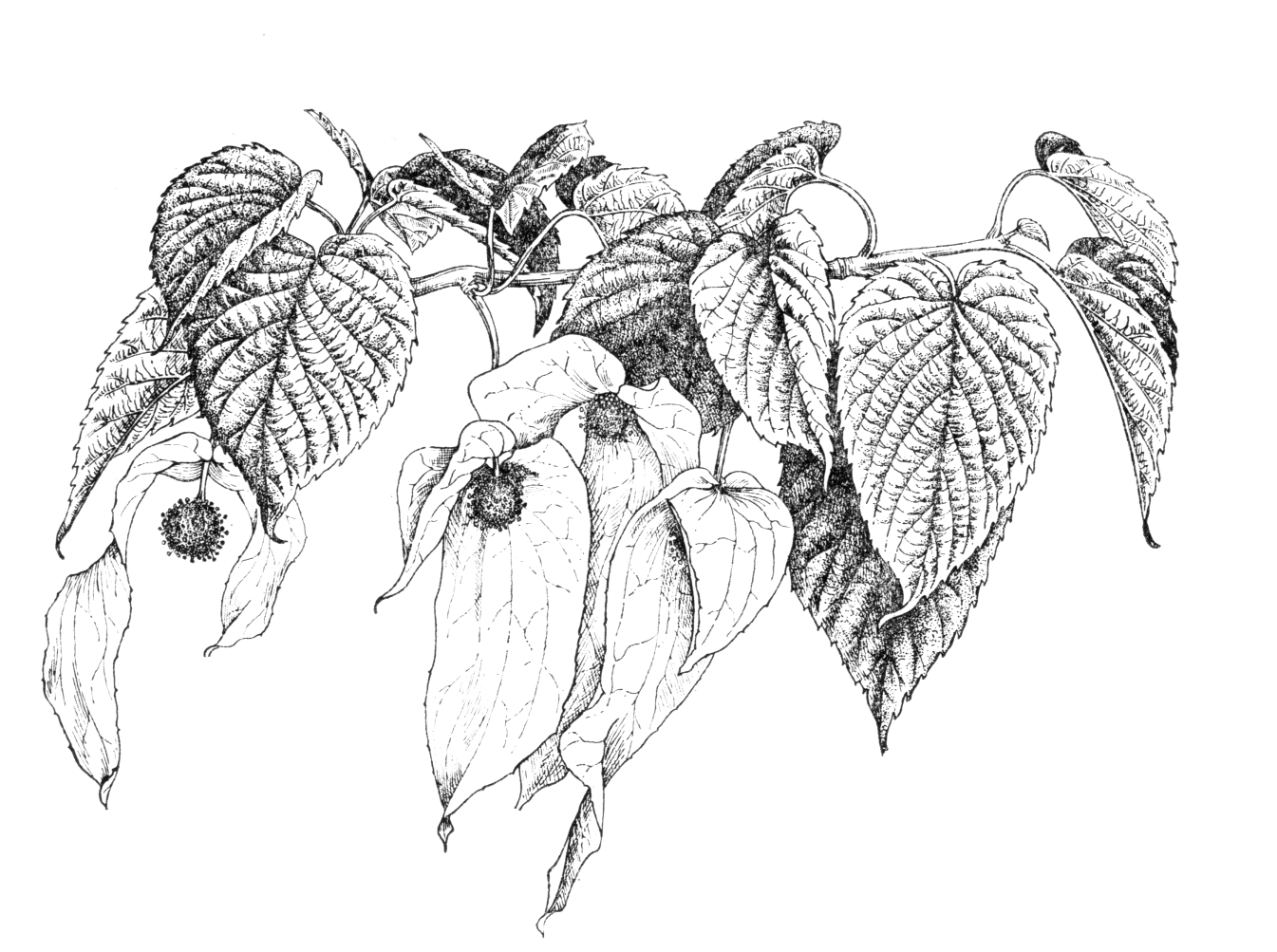
Deciduous tree to 20 m with grey-brown bark, becoming flaky with age, the crown shape often likened to a lime (Tilia). Branchlets glabrous, with a glaucous bloom at first. Leaves also resembling a lime, alternate, 8–15 × 7–12 cm, ovate, base cordate, margin serrate, apex acuminate, the upper surface silky-hairy, the lower surface sometimes pubescent. Petiole red, 4–7 cm, stipules absent. The young foliage is scented. Inflorescence globose, purple in bud, 2 cm across on a drooping 7 cm peduncle, with two (rarely three) conspicuous, ovate, opposite, unequal bracts to 16 cm and 8 cm respectively. Bracts overhanging the inflorescence, green, becoming white at floral maturity. The inflorescence consists of many very small flowers, all lacking a perianth: all but one is male, with one to seven stamens with reddish anthers; there is a single hermaphrodite flower with inferior ovary, style short and stout, 6–10 lobed, with inflated stigmas. The whole is arguably a structure intermediate between flower and inflorescence. Fruit a drupe, 3–4 × 1.5–2 cm, ovoid, oblate or pear-shaped, purplish-green maturing to pale brown. A fleshy mericarp surrounds the ridged, woody endocarp. Flowering time April (wild trees), late April to May (UK). (Claßen-Bockhoff & Arndt 2018; Sun & Huang 2011; Qin & Phengklai 2007; Bean 1981).
Distribution China Guizhou, W Hubei, W Hunan, Sichuan, N Yunnan
Habitat Montane mixed forests; 1100–2600 m.
USDA Hardiness Zone 6-9
RHS Hardiness Rating H5
Awards AGM
Conservation status Not evaluated (NE)
A mature Handkerchief Tree in full flower is a fine sight. So desirable that the sole aim of E.H. Wilson’s first expedition to China was to collect it, so exotic that during its early years in Britain the Times newspaper announced flowerings at Kew, as a representative of the Chinese flora in western gardens it is nothing less than iconic. Even its perception in China is coloured by its status in foreign gardens. Chinese political leaders have been impressed by flowering specimens when visiting both Geneva and Washington, DC (Sun & Huang 2011), while a specimen of the precociously flowering cultivar ‘Sonoma’ featured in a Chinese television programme on Chinese plants in western gardens (Lancaster 2019).
It is the large white bracts, contrasting with fresh young foliage, which make Davidia special. It has long been assumed that they attract pollinating insects. Once it became clear that they hang down, the possibility that they might act as umbrellas, reducing pollen loss to rain, also presented itself. Experiments in the wild (Sun et al. 2008; Sun & Huang 2011) comparing pollinator visits and pollen loss between intact inflorescences, those with bracts removed and others with the bracts replaced by green or white paper look-alikes, lent support to both these hypotheses. The major pollinators are bees, which are attracted only once the bracts turn white. In British gardens it is visited by various bee species (pers. obs.); a big tree in flower gives off a ‘buzz’ in the manner of a flowering lime. The mass of staminate flowers generally appears dark, contrasting with the white bracts, but occasionally a ‘blonde’ form is found in which the flower cluster lacks dark pigments and appears green (pers. comm. John Grimshaw 2019).
Davidia involucrata was discovered by Armand David in 1869, in Baoxing County, Sichuan and described by Baillon (1871) from his material. The story of its introduction is one of rivalry between international interests (Kilpatrick 2014). With all the early running made by Frenchmen, Adrien Franchet working at the Muséum national d’Histoire naturelle in Paris was keen for the first introduction of live material also to be a French one. At his behest, the missionary Paul Farges collected seed in 1896, supplying both Franchet and Maurice de Vilmorin. The latter established a single seedling, from which material propagated by cuttings was distributed internationally even before its first flowering in 1906. Further seed followed. Anglo-Saxon efforts were belated, but on an altogether grander scale. English nurseryman Harry Veitch commissioned E.H. Wilson to travel to China in 1899 with the sole aim of collecting this species. Ignorant of Farges’ collection, and directed to likely areas by Augustine Henry, Wilson collected over 14,000 seeds (W 930) in 1900, near Yichang, Hubei: Veitch raised over 13,000 seedlings, forming the backbone of our cultivated stock (Howard 1980). Given its desirability, one might have expected this to result in near-ubiquity in young plantings within a few years, yet this was not the case. It seems that Veitch made a commercial decision to destroy a large proportion of the stock, calculating that more profit could be made by pricing just a few hundred trees as rarities (Gordon 2021). While several thousand were burnt, more were saved through ‘timely action’, Wilson (1917) later describing Veitch’s ploy as ‘a whim of an unbalanced mind’.
In Wilson’s subsequent encounters with Davidia he never saw this copious seed set, and his few later collections were small and less influential (perhaps concentrating his bitterness at Veitch’s earlier wastefulness). At a population level, low seed production is compounded by short-range dispersal and high seedling mortality: Su & Su (2005) concluded that in a Wolong population, most individuals were the result of sprouting from the bases of fallen trees. Wickenden & Lear (2009) found the same in a population in the Upper Dulong Valley of Yunnan, which proved to be a westerly extension to the known range of the species. They also repeat an interesting but unattributed claim that flying foxes take the fruits, and that Davidia groves are often flying fox roosts. It is easy to speculate that changes to roosting patterns could leave an established Davidia population with no effective means of seed dispersal. Vegetative replacement of fallen trees is also seen in cultivation: a mature specimen at Dartington Hall, Devon, is the result of sprouting from the base of a tree planted in 1915 and was ‘magnificent’ by the 1960s (pers. comm. T. Underhill), which was brought down by heavy snow in 1969 (Snell 1989). Vigorous young shoots are often to be seen from the base of the trunk of even quite mature trees, giving no doubt about its potential for coppice regeneration.
Davidia seems to tolerate a wide range of soils, thriving even on chalk at Highdown, Sussex (Stern 1974). A sheltered position is desirable however, as it is susceptible to spring frosts, and it will flourish best in good soil.
With both supply and demand being high from the start, large specimens are widespread in Europe and suitable parts of North America. The tallest British specimen (23.5 m in 2014) is at Weasenham Woods, Norfolk, while the British champion by girth (98 cm diameter, 2014) is at Tregrehan, Cornwall (The Tree Register 2019). The Dove Tree is widely grown across France, the Low Countries, Germany and northern Italy; a good specimen has been noted as far east as the Arboretum Mlynany, Slovakia (monumentaltrees.com 2018). In North America, it has proved successful along the eastern seaboard at least as far north as the Boston, MA area, with old specimens from both Farges and Wilson collections at the Arnold Arboretum (Arnold Arboretum 2018). Good specimens are also seen west of the Cascade – Sierra Nevada axis as far north as the Vancouver area of British Columbia, in Washington Park, Seattle, for example (Jacobson 1996). Away from these areas, it is at best a subject for optimistic experimentation in North America, deserving of a sheltered position.
The wildly different germination success of early introductions points to a problem relevant both to horticulture and conservation, for despite not being assessed by the IUCN, Davidia is widely considered in Chinese literature (e.g. Fu & Jin 1992) as being under threat. Fruits contain a variable number of embryos, reduced by abortion during fruit development, apparently an active response to a tree’s external and internal environment (Li et al. 2016). The established picture is of double dormancy, with the radicle emerging after a period of warmth, but further growth delayed until after exposure to cold (Dirr 2009). It is not at all clear whether all the viable embryos germinate at once. It may be sufficient to pack newly collected fruits into a large pot of leaf mould left outside over two winters, anything germinating being removed and potted up in the second spring (McAllister & Burston 1987). Where multiple embryos germinate (these authors report anything up to six seedlings from a single fruit) they can be separated and potted individually. This technique relies on sufficiently cold winters, and is not always effective in southern England (pers. comm. D. Joyce). A more controlled regime involves storing seeds packed in damp compost for 12–20 weeks at 15–20 °C; those with a radicle visible are then kept at about 4 °C for 12–16 weeks (Tree Seed Online Ltd 2018). Seedlings seem very intolerant of drought or waterlogging, and root growth is slow: Hugh McAllister (pers. comm.) finds results are best in a coarse peaty compost, in small but frequently watered pots. Despite these insights, germination remains somewhat unpredictable, and the mechanisms underlying dormancy are the subject of ongoing research.
Our understanding of variation within this species has not been helped by the early publication of two botanical varieties, v. vilmoriniana (Dode) Wangerin with the leaf undersides glabrous, or pubescent only when young, and the type v. involucrata with densely pubescent undersides – and perhaps a degree of nationalism. Both the Farges and the vast 1900 Wilson collections were claimed as v. vilmoriniana, while two of Wilson’s later collections from 1903 and 1904 were labelled v. involucrata. Later authors (for example Bean 1981) agreed that the bulk of cultivated material was v. vilmoriniana, whether or not labelled as such. Ames (1931) was already casting doubt on the botanical and horticultural validity of these varieties, whose defining characters do not correlate closely with any others, which sometimes occur together in wild populations, and which seem to be linked by intermediates. Attempts have been made to correlate fruit shape with variety, those of v. vilmoriniana being more elongated; Wickenden & Lear (2009) found both fruit shapes in a single Yunnan population, and exceptions have been found even within the limited range of cultivated material (Bean 1981). For a detailed treatment of the uncertain nature of the horticultural evidence, see Bean (1981) and Clarke (1988). Persistent claims that v. vilmoriniana is the hardier may indicate that variation exists, but does not itself support the existence of these varieties, as provenance may have an important effect on hardiness. A claim repeated online (for example Highdown Gardens 2019), always without a supporting reference, that the two varieties have different chromosome numbers and cannot produce fertile hybrids, is most likely merely an extrapolation from an inconsistency between early chromosome counts, nicely cleared up by the thorough karyological work of He et al. (2004). To persist with these varities would be to prolong our collective agony and we treat them here as a single entity, Davidia involucrata.
The selection and propagation of Davidia cultivars is a recent phenomenon, with Japan and Oregon as centres of activity. At the time of writing (2019) only ‘Sonoma’ has had anything approaching a wide distribution in Europe or North America. Propagation is generally by grafting onto seed- or cutting-raised D. involucrata: Oregon nurseryman Talon Buchholz (2013) cites availability of stocks rather than scion wood as limiting commercial propagation. ‘Sonoma’ at least is also available from tip cuttings. Cultivar information from Hatch et al. (2017), Buchholz & Buchholz Nursery (2019) and Buchholz (2013).
'Aya Nishiki'
Variegated foliage: yellow marginal or submarginal band, leaves tending to scorch in dry-summer air. Origin: Japanese nurseryman, Akira Shibamachi, as aseedling. Oddly, the plant patent application describes a cream margin, while all commercial descriptions and photographs appear to show an unambiguously yellow margin: Dirr (2009) explains this by saying the variegation diminishes later in the season, but also says it is cream. It is probable that the clone circulating as ‘Shibamichi’ is the same: Dirr reports that although Akira Shibamichi’s clone was patented by Hines (US PP17066) it was independently imported to the United States by other nurseries.
'Crimson Spring'
Burgundy-red new growth, ageing bronze-red then green. A US plant patent (17203P3) was granted to ‘Purpleleaf’, another purple-leaved cultivar selected by A. Shibamachi as a seedling; this appears to have had little or no commercial distribution (2019). Dirr (2009) tartly notes that the claim that the colour persists through the growing season is ‘patentedly [sic] untrue’.
'Iseli Fastigiate'
Narrowly erect, selected as a seedling at Iseli Nursery, Oregon; introduced 2013. Other narrow-crowned forms include ‘Columna’, perhaps also slower growing, originating at the Italian nursery Gheri Vivai Piante (Havlis 2019) and ‘Kylee’s Columnar’.
'Lady Dahlia'
Variegated foliage: leaves very irregularly centred, yellowish-green to chartreuse, sometimes claimed as ‘gold’. A specimen can be seen at the JC Raulston Arboretum, North Carolina.
'Lady Sunshine'
Variegated foliage: broad, irregular yellow margin, maturing to cream; distinct from ‘Aya Nishiki’, which shows submarginal banding. Selected by Oregon nurseryman Crispin Silva. A specimen was planted in the Arnold Arboretum in 2018 (Arnold Arboretum 2018).
'Platt's Variegated'
Variegated foliage irregularly mottled and sectored white. Selected as a seedling at the Platt Garden, Portland, Oregon, the original is now a sizeable tree.
'Shibamachi Variegated'
Variegated foliage: broad yellow margin, red-tinged edge in autumn. Probably the same as ‘Aya Nishiki’, q.v.
'Sonoma'
Flowers well at a young age, even on young grafted specimens about 30 cm tall; bracts larger (to 25 cm long) than in many other cultivated davidias. Roy Lancaster (2019) describes a young, 2 m tall specimen in his own garden bearing about 100 inflorescences. One of the first cultivars, it originated at the Sonoma Horticultural Nursery, California, before 1984. Its floral precocity revolutionises Davidia as a garden tree, making it possible for everyone to grow a flowering tree. Handkerchief Trees are known to be able to sprout from the base: precocious flowering in ‘Sonoma’ opens the way for an experimentally-minded gardener to attempt a coppiced, shrubby specimen, which would suit a small garden for the long term. It would be important to choose a tree propagated from a tip cutting for this experiment.
'Sumako'
Purple leaves with yellow-green veins.
'White Dust'
Variegated foliage: irregular white and pale green spotting, without striking white sectors. Japanese origin.