Styrax
Sponsor
Kindly sponsored by
Arabella Lennox-Boyd
Credits
John Grimshaw, Ross Bayton & Alan Elliott (2017)
Recommended citation
Grimshaw, J., Bayton, R. & Elliott, A. (2017), 'Styrax' from the website Trees and Shrubs Online (treesandshrubsonline.
Family
- Styracaceae
Common Names
- Snowbells
- Storax
Species in genus
- Styrax americanus
- Styrax calvescens
- Styrax confusus
- Styrax dasyanthus
- Styrax faberi
- Styrax formosanus
- Styrax grandifolius
- Styrax hemsleyanus
- Styrax hookeri
- Styrax japonicus
- Styrax limprichtii
- Styrax obassia
- Styrax odoratissimus
- Styrax officinalis
- Styrax platanifolius
- Styrax redivivus
- Styrax serrulatus
- Styrax shiraianus
- Styrax suberifolius
- Styrax tonkinensis
- Styrax wilsonii
- Styrax wuyuanensis
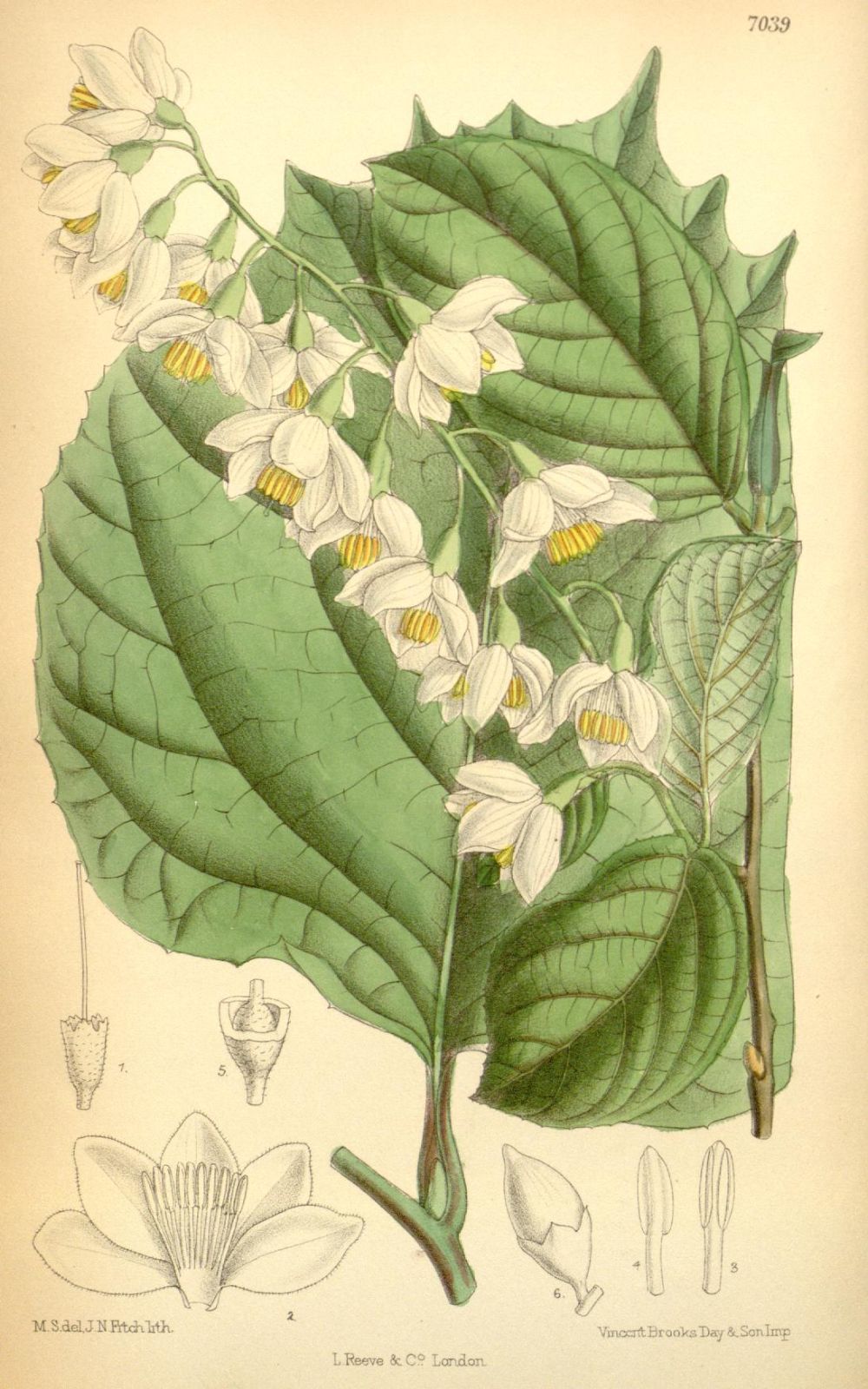
There are about 130 species of Styrax, occurring in eastern Asia, North and South America and the Mediterranean. They are evergreen or deciduous trees with several winter buds at each node, each with one outer scale. The leaves are alternate, petiolate, glabrous to stellate and with sub-entire to serrate margins; they often vary greatly in size on the shoot, the uppermost frequently being much larger than those below. Inflorescences are axillary or terminal, solitary or several per fascicle; individual inflorescences may be racemose, cymose, paniculate or even single-flowered. The flowers are hermaphrodite and typically 5-merous; the calyx cup-like, with (two to) five (to seven) teeth or lobes, the corolla white or pink, campanulate, 5– to 7-lobed, the lobes reflexed or not; with (8 –)10(–13) stamens, usually all of equal length. The fruit is a fleshy drupe or a dry capsule, which may be dehiscent (and then three-valved) or indehiscent; with one (to two) hard seeds per locule (Gonsoulin 1974, Spongberg 1976, Hwang & Grimes 1996, Fritsch 1997, Huang, Fritsch & Shi 2003).
Styrax, together with other members of the Styracaceae, is the subject of ongoing scientific research by Peter W. Fritsch and his associates, based at the California Academy of Sciences, San Francisco. Their work has greatly improved our understanding of the genus and family (Fritsch et al. 2001; Huang, Fritsch & Shi 2003; Fritsch 2004); where possible, the accounts below follow their views.
Styrax – described by Tom Hudson (2004) as having ‘poise and style’ – is an up-and-coming genus in horticulture, many newly introduced species having recently joined the rather few that are well known. Of these, only S. japonicus is really widely grown, and this is the only species in which cultivars have been selected. Amongst the varied assortment of cultivars with different growth habits or flower colour, ‘Sohuksan’ (syn. ‘Emerald Pagoda’), selected by J. C. Raulston from seedlings of a collection made in South Korea in 1985, is generally acclaimed to be the finest, having larger flowers than normal, and larger, darker leaves, and seeming to be more heat-tolerant than others. Dirr (1998) considered it to be one of Raulston’s best introductions. ‘Crystal’ came from the same collection. Styrax grandiflorus Griff. is a synonym of S. japonicus, occasionally found on labels. Styrax obassia is also quite frequently grown, especially in North American collections (P. Fritsch pers. comm. 2008), but most others are rather rare.
The pendulous, usually five-pointed, usually white corollas are very characteristic of the genus, and have given rise to the popular name ‘snowbells’, especially for S. japonicus. Styrax in general seems to be quite variable, and varieties have been recognised in several of the species described in the present work. Significant ones are mentioned briefly below. There is much local endemism, especially in the southeastern United States and adjacent Mexico (from where future introductions might be expected), as well as in populations stretching into South America (Fritsch 1997). The growth habit varies considerably, from quite small and shrubby to trees of a size useful in forestry, such as S. tonkinensis. (As the genus is so scantily known in horticulture, we bend our usual rules slightly here to include descriptions of some of the smaller species.) After much discussion Styrax is now accepted (Nicolson & Steyskal 1976) to be of masculine gender, resulting in most names ending in –us, rather than the familiar feminine -a or the occasionally seen neuter -um.
Most arboreta have a small representation of Styrax, but a comprehensive assortment is harder to find. J. C. Raulston built up a good collection, which he wrote about (Raulston 1991; Raulston 1992), and the current plantings at the JC Raulston Arboretum remain reasonably diverse. On the West Coast the University of California Botanical Garden at Berkeley and the San Francisco Botanical Garden grow a selection of taxa, including some Mexican species apparently not much grown elsewhere. Further north, the University of British Columbia Botanical Garden has an interesting assemblage of Asian species, and these also feature strongly in private collections in southwestern England, such as that of Tom Hudson at Tregrehan, and at Gwavas Station, the collection of Mike and Carola Hudson in Hawkes Bay, New Zealand. Many Styrax are tolerant of less benign climates and many will perform well in apparently less favoured locations. Remarkably, there are two National Plant Collections of Styracaceae in the vicinity of Morecambe Bay in northwest England, an area with cool summers and high rainfall, but comparatively mild winters; both encompass the full range of the family and contain thriving examples of most genera in cultivation. These are at Holker Hall, Cumbria, where the collection has been built up by the Cavendish family, and at Gresgarth Hall, Lancashire, where Arabella Lennox-Boyd has planted them extensively in her woodland garden. She has kindly sponsored the production of entries for the family for Trees and Shrubs Online.
None of the essentially Mexican taxa, or those from further south in Latin America, are widely grown in our area. Styrax platanifolius, with two subspecies in cultivation, is described below. Styrax ramirezii Greenm., potentially a tree to over 20 m, with a wide range in Mexico, is grown in the San Francisco Bay Area but is probably not hardy further north; it has large evergreen leaves and smallish flowers, of white or pink. Styrax glabrescens Benth., ranging from northeastern Mexico to Costa Rica, is another evergreen, with trees to 30 m. It survived in the JC Raulston Arboretum for 10 years until lost to a late freeze (T. Lasseigne pers. comm. 2007), and has grown in Berkeley since 1991. Raulston (1992) recorded that a mysterious ‘S. globosus’ was being cultivated by Yucca Do Nursery in Texas, but this was in fact S. lanceolatus P.W. Fritsch (P. Fritsch pers. comm. 2008). It is not known if it has persisted in cultivation.
Several poorly known Asian species are also grown in our area, including S. macrocarpus W. C. Cheng from Guangdong and Hunan, which has large leaves uniquely preceded by solitary flowers opening on the bare branches – although this has not done well at Tregrehan, and probably requires greater warmth in summer. The smaller S. rugosus Kurz, from India, Myanmar and Yunnan, also attempted at Tregrehan, is another that has not been successful there. Material grown as S. huanus Rehder is more promising. The taxon has been sunk into S. hemsleyanus (Huang, Fritsch & Shi 2003), differing from it principally in being heavily pubescent, but otherwise falling completely within the range of variation expected in a Styrax species (P. Fritsch pers. comm. 2008). At his home in Cornwall the late Edward Needham had a fine specimen labelled S. huanus, collected by himself in China, and propagations from this are being grown elsewhere in that county. It is a ‘good thing’, according to Tom Hudson (pers. comm. 2007).
If the example of the few familiar species can be used as a guide, cultivation requirements for the genus include warmth but not great heat, shelter but not deep shade, and a moist acidic soil. In the British Isles summers tend to be on the cool side for them, and they need as much warmth as possible. Some, including S. japonicus, can tolerate full sun in cooler climates, but in general they are prone to sunburn where light intensity is too great. In eastern North America light shade is advised for S. japonicus in areas covered by Zones 7–8 (Dirr 1998), and the heat and humidity of the lowland southeastern United States are too much for most potentially hardy species (Raulston 1991). In all areas most require ample summer moisture, though taxa from Mediterranean climates need a dry summer. Propagation is by cuttings taken in early summer, but it can be difficult to get them through the following winter. The seed has a double dormancy so patience is required (Huxley, Griffiths & Levy 1992; Dirr 1998).
The key to cultivated species has been developed from the following sources Hwang & Grimes 1996; Fritsch 1997; Grimshaw & Bayton 2009; Lobdell 2013.
Identification key | ||
| 1a. | Plants evergreen. | Styrax suberifolius |
| 1b. | Plants deciduous. | 2. |
| 2a. | Inflorescence strictly terminal; leaf margin entire, lobed or coarsely toothed, never glandular dentate or glandular serrate. | 3. |
| 2b. | Inflorescence lateral or terminal (often reduced to single or few-flowered) leaf margin on sterile shoots glandular dentate or glandular serrate. | 5. |
| 3a. | Stellate hairs of sterile branches and leaves white or light brown, pedicel 8–17mm and uniform in thickness, pollen pale yellow. | Styrax officinalis |
| 3b. | Stellate hairs of sterile branches and leaves yellow to dark brown, pedicel 4–9mm and tapering from apex to base, pollen orange or bright yellow. | 4. |
| 4a. | Larger undehisced capsules 7–10 mm, hairs on capsule greyish; pedicel 1.3–2.3 times length of calyx; leaves often coarsely lobed or irregularly undulate. | Styrax platanifolius |
| 4b. | Larger undehisced capsules 11–15 mm, hairs on capsule brownish; pedicel 0.5–1.4 times length of calyx | Styrax redivivus |
| 5a. | Corolla lobes margins narrowly curved or rolled inward but never overlapping. | 6. |
| 5b. | Corolla lobes never curved or rolled inward, overlapping. | 12. |
| 6a. | Calyx and pedicel glabrous. | Styrax wuyuanensis |
| 6b. | Calyx and pedicel densely scaly or stellate pubescent. | 7. |
| 7a. | Underside of leaf densely stellate pubscent. | Styrax calvescens |
| 7b. | Underside of leaf glabrous or sparsely stellate pubscent. | 8. |
| 8a. | Fruit cylindric to oblique ovoid or ellipsoid to ellipsoid-ovoid; filaments flexuous. | Styrax serrulatus |
| 8b. | Fruit subglobose, globose, ovoid, or obovoid; filaments straight. | 9. |
| 9a. | Leaves leathery or subleathery; inflorescences (3–)5-flowered or more. | 10. |
| 9b. | Leaves papery; inflorescence to 5-flowered. | 11. |
| 10a | Inflorescences racemose; fruit 8–15 mm in diameter. | Styrax confusus |
| 10b | Inflorescences panculate; fruit 5–7 mm in diameter. | Styrax dasyanthus |
| 11a. | Calyx 2.5–3 mm; fruit irregularly wrinkled. | Styrax formosanus |
| 11b. | Calyx 4–5 mm; fruit smooth. | Styrax faberi |
| 12a | Pseudoterminal inflorescences ≥ 7 cm long, often 8–23-flowered. | 13. |
| 12b | Pseudoterminal inflorescences < 7 cm long, ≤ 7-flowered (3–11-flowered in S. shiraianus). | 18. |
| 13a. | Lowest leaves on shoots alternate. | 14. |
| 13b. | Lowest leaves on shoots opposite or subopposite. | 15. |
| 14a. | Lower leaf surface not visible through tomentum; filaments of equal width throughout. | Styrax tonkinensis |
| 14b. | Lower leaf surface visible through the pubescence, if present; filaments narrowing from base to apex. | Styrax odoratissimus |
| 15a. | Petiole base of larger leaves swollen to cover bud. | Styrax obassia |
| 15b. | Petiole base of larger leaves not swollen, not covering bud. | 16. |
| 16a. | Vegetative end buds 4–6 mm long; calyx narrowly cup-like, fruit globose to ovoid, dehiscent. | Styrax hemsleyanus |
| 16b. | Vegetative end buds ≤ 3 mm long; calyx campanulate or broadly cup-like; fruit globose, indehiscent or rarely dehiscent. | 17. |
| 17a. | Tree not suckering from roots; corolla lobes 11–23 × 6–10 mm; fruit 10–17 × 9–19mm. | Styrax glabrescens |
| 17b. | Tree often profusely suckering from the roots; corolla lobes 8–16 × 3–7 mm; fruit 8–12 × 6–8 mm. | Styrax grandifolius |
| 18a. | Petiole of larger leaves swollen at base, covering the bud. | Styrax shiraianus |
| 18b. | Petiole of larger leaves not swollen at base, not covering the bud. | 19. |
| 19a. | Calyx truncate, undulate, irregularly lobed or toothed, if toothed then the teeth not touching; calyx abaxially glabrous, or if stellate trichomes present, within 1 mm of the margin more sparsely pubescent than the rest of the calyx or subglabrous to glabrous. | 20. |
| 19b. | Calyx distinctly dentate, the teeth usually touching or separated by a shallow concave portion; calyx abaxially within 1 mm of the margin evenly pubescent. | 22. |
| 20a. | Longer pedicels on each twig 15–50 mm long, usually equal to or longer than subtended flower. | Styrax japonicus |
| 20b. | Longer pedicels on each twig 2–10(–13) mm long, usually shorter than subtended flower. | 21. |
| 21a. | Stems of young fertile shoots generally < 0.6 mm wide at the narrowest points toward the base; pedicels 0.2–0.6 mm wide at the base; corolla lobes 1–5 mm wide, apex acute. | Styrax americanus |
| 21b. | Stems of young fertile shoots generally 1 mm wide at base but often narrower towards apex); pedicels 0.6–1 mm wide at base; corolla lobes 3–13 mm wide, apex obtuse or acute-acuminate. | Styrax hookeri |
| 22a. | Leaves 1–2.5(4) × 0.7–2(2.5) cm; fruit 0.4–0.6 cm wide. | Styrax wilsonii |
| 22b. | Leaves 3–13 × 2–8 cm; fruit ≥ 0.7 cm wide. | Styrax limprichtii |